UKM Hospital To Conduct Study On Potential Therapy For Leukaemia

By Durratul Ain Ahmad Fuad
KUALA LUMPUR (Bernama) – A potentially effective treatment for patients with relapsed and refractory acute lymphoblastic leukaemia (ALL) is in the works.
ALL is a type of blood cancer in which the bone marrow makes too many lymphocytes, a type of white blood cell.
Universiti Kebangsaan Malaysia’s teaching facility Hospital Canselor Tuanku Muhriz (HCTM) is collaborating with Plutonet Sdn Bhd on a study and clinical trial involving the Chimeric Antigen Receptor-T Cell (CAR-T) therapy, a revolutionary treatment found to be effective for ALL patients who cannot be treated with chemotherapy or are immune to it.
Under an agreement signed last month between HCTM’s Cell Therapy Centre and Plutonet – which specialises in cell and gene immunotherapy – 10 patients are set to participate in Malaysia’s first-ever clinical study involving the CAR-T cell therapy.
The HCTM-Plutonet collaboration will focus on research and development of CAR-T cell treatment technology. The clinical study, for which Plutonet is contributing RM1 million, will look into the effectiveness and safety of the CAR-T cell therapy.
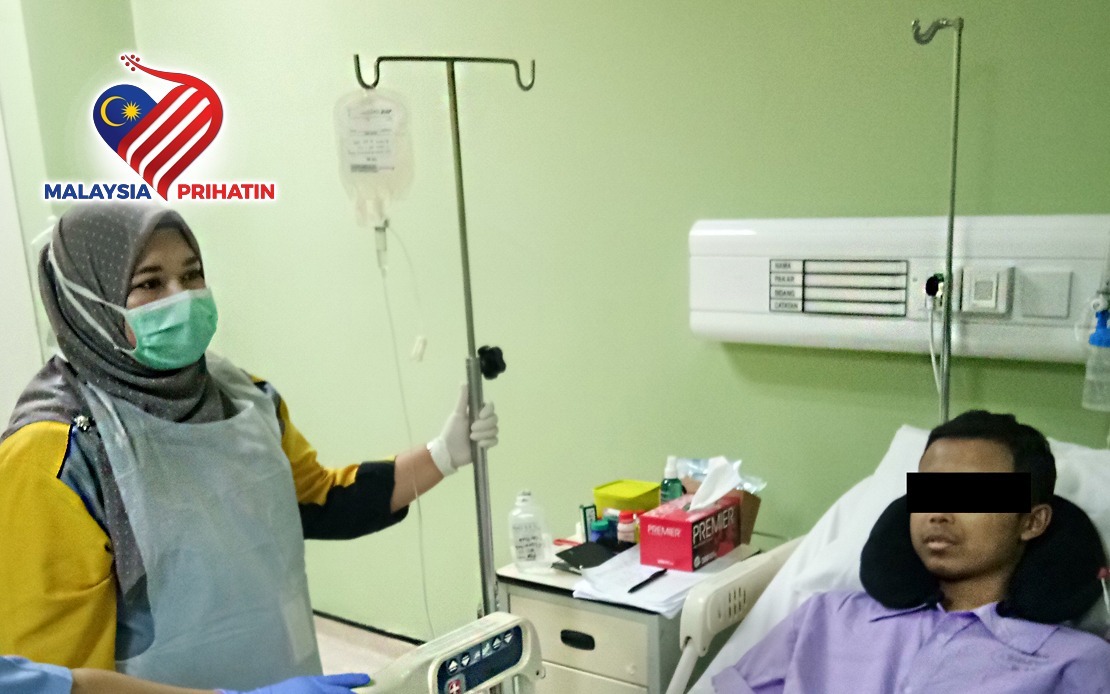
The CAR-T cells are induced into the patient through intravenous transfusion. -- Photo courtesy of Universiti Kebangsaan Malaysia’s teaching facility Hospital Canselor Tuanku Muhriz (HCTM)
Head of HCTM’s Cell Therapy Centre Prof Dr S Fadilah Abdul Wahid said for their CAR-T cell therapy study, her team will be using the CD19-directed CAR-T cells that can identify and kill cancer cells.
(CD19 is a protein that activates the necessary antibodies when injected into the body while T cells are one of the major components of the immune system.)
The CAR-T cell therapy involves the use of genetically modified T cells which after it is transfused into the patient has the ability to target and kill cancerous cells.
“For now, this therapy is the last option for (blood cancer) patients. If they have problems with chemotherapy, they can try this therapy,” Dr S Fadilah, who is also the chief researcher for the HCTM-Plutonet study, told Bernama in an interview.
PROCEDURES
pic-3
CAR-T cell therapy was first developed in 1993 by Zelig Eshhar from Weizmann Institute in Israel and many years later Prof Carl June from the University of Pennsylvania in the United States tested it further to prove its efficacy.
In early 2017, the United States Food and Drug Authority approved the use of CD19 CAR-T cell therapy for ALL patients. The therapy has also been approved by the European Medicines Agency, as well as Australian and Japanese regulators.
Data from 2017 showed that there was a 70 percent probability of achieving complete remission (no evidence of leukemia) for patients who underwent CAR-T cell therapy, according to Dr S Fadilah.
“Based on the findings, the treatment is believed to be able to save lives,” she said, adding that ALL patients aged 13 and above can opt for CAR-T cell therapy after they are screened by the hospital.
Explaining how the therapy is carried out, she said the patient first has to undergo a three- to four-hour-long procedure called leukapheresis to extract mononuclear cells from the patient’s blood.
The extracted mononuclear cells are then sent to a specialised laboratory where the CAR-T cell production and culturing process takes place over 10 to 21 days.
Prior to transfusing the CAR-T cells into the patient, they have to go for a bone marrow test to determine the level of cancerous cells and undergo low-dose chemotherapy for three days.
“The CAR-T cells are induced into the patient only once through intravenous transfusion,” said Dr S Fadilah, adding that the effects of the treatment can be seen within three to four weeks.
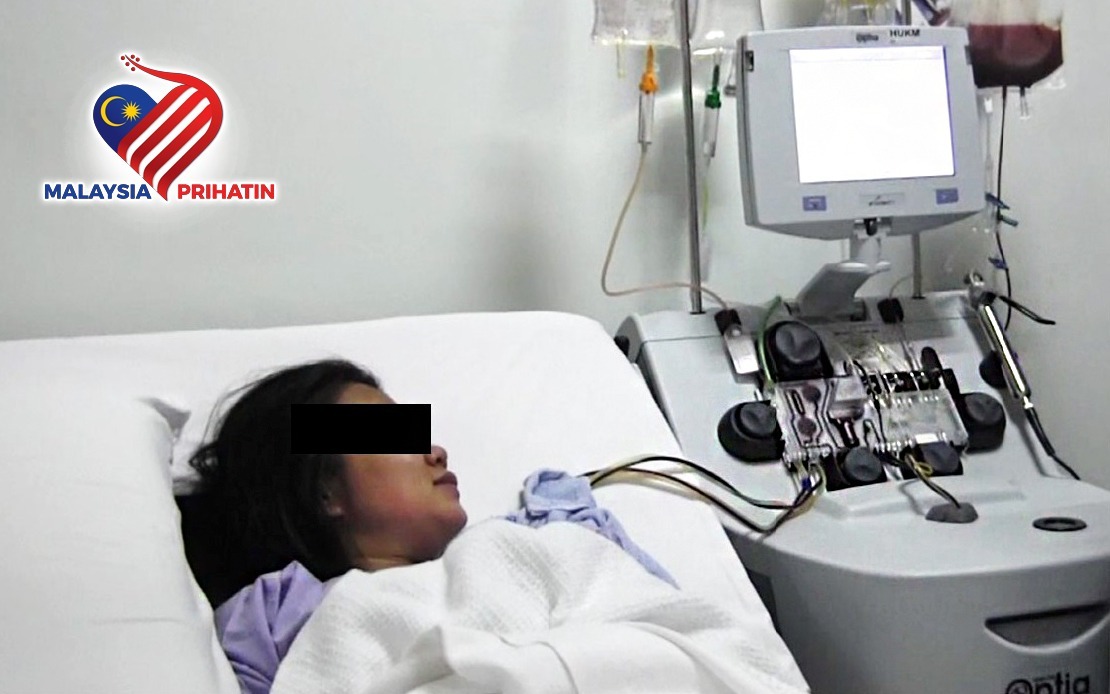
A patient undergoing treatment at HCTM's Cell Therapy Centre. -- Photo courtesy of Universiti Kebangsaan Malaysia’s teaching facility Hospital Canselor Tuanku Muhriz (HCTM)
SIDE-EFFECTS
pic-2
She said the CAR-T cell treatment will take about a year to complete while full recovery will take about five years.
However, she added, three to four weeks after the CAR-T cell infusion, the patient may have to contend with two “side-effects”, namely cytokine release syndrome (CRS) and neurotoxicity.
CRS is an acute systemic inflammatory syndrome characterised by fever and multiple organ dysfunction while neurotoxicity refers to damage to the brain or peripheral nervous system caused by exposure to natural or man-made toxic substances.
“CRS and neurotoxicity are the challenges patients who undergo CAR-T cell therapy have to face. However, these two side-effects can be treated. Once the patients go past this stage, they will gradually recover,” said Dr S Fadilah.
According to data from the Malaysian National Cancer Registry Report 2012-2016, leukaemia was the sixth most common cancer among adults and most common cancer among children in Malaysia.
HCTM receives about 25 cases of child leukaemia patients every year, added Dr S Fadilah.
Translated by Rema Nambiar
BERNAMA
For more information click here
HealthEdge
EXCLUSIVE

Pet Vaccination, Public Awareness And Surveillance Key Towards Rabies-free Southeast Asia - Experts
KUCHING, Dec 11 (Bernama) -- The goal of making Southeast Asia free from human rabies can be achieved through a total understanding of the disease, how it can be prevented and responsible pet ownership among communities, say experts.
read more ››IN FOCUS

TAVI KAEDAH BAIK PULIH INJAP JANTUNG TANPA PEMBEDAHAN



